

Sphalerite occurs in many colors. With a dispersion over three times that of diamond and an adamantine luster, faceted specimens make beautiful additions to gem collections. However, they're too soft for most jewelry uses.
4 Minute Read
Radiant square-cut sphalerite, 198 cts, 30.1 x 30.3 x 21.1 mm, Las Manforas, Aliva, Picos de Europa, Spain.
Sphalerites have low hardness (3.5 to 4) and perfect cleavage, which make them less than ideal as jewelry stones. Nevertheless, sphalerites can show beautiful colors and may appeal to collectors of unusual gems. Use protective settings for these gems, especially if worn as ring stones.
This 18K gold cocktail ring setting allows light to enter the 35.7-ct sphalerite center stone but still protects it from scratches and blows. The ring also features pavé diamonds and tiny sphalerites. Photo courtesy of liveauctioneers.com and I.M. Chait Gallery/Auctioneers.
For custom cutting, you can easily find facetable sphalerite rough in most colors. Faceting this material, however, poses a challenge. Consult our sphalerite buying guide for more information.

Sphalerites come in many colors, including brown, orange, yellow, green, orange-red, red, whitish gray, black, and even (very rarely) colorless. Sphalerites can also show color zoning.

According to a 1984 study by Hoffmann and Henn, a number of trace elements may contribute to color in sphalerites. These include germanium, calcium, copper, mercury, and cerium (yellow); tin, silver, and molybdenum (reddish); and cobalt and iron (green). (p. 49) Iron-rich sphalerites appear black.

Iron-rich, opaque, black stones are known as marmatites, named after the locality, Marmato, Italy.
In Europe, sphalerite is sometimes called blende, after the German word blenden, "to dazzle." Though they have no connection to actual ruby gems, stones with red and orange shades are sometimes called "ruby blende."

Low-iron, pale and colorless sphalerites, known as cleiophanes, are extremely rare.

Sphalerites have an exceptionally high dispersion of 0.156. Well-cut sphalerites can display a beautiful rainbow effect of multi-colored flashes of light. Only other rarely faceted gemstones have comparable dispersion values. Gemologists can measure a gem's dispersion using a refractometer or a spectrometer. However, keep in mind that cut quality, color, and specific gravity can affect how well a specimen displays dispersion.

Sphalerite and wurtzite are polymorphs. They share the same chemical formula (ZnS), but have different crystal habits. Sphalerite has an isometric crystal structure, while wurtzite has a hexagonal structure. These two minerals can coexist in alternating layers as schalenblende, a gem material used rarely for cabochons.

Sphalerites may have a streak color ranging from pale brown to yellow or white. Keep in mind that streak testing can destroy the test sample. Conduct this examination only as a last resort for gem identification on rough, never on a finished gem. This test will also release a sulfurous odor from the test sample.
Sphalerites are pyroelectric. When heated, these gems generate an electrical charge.
Sphalerites may show anomalous double refraction due to crystal strain.
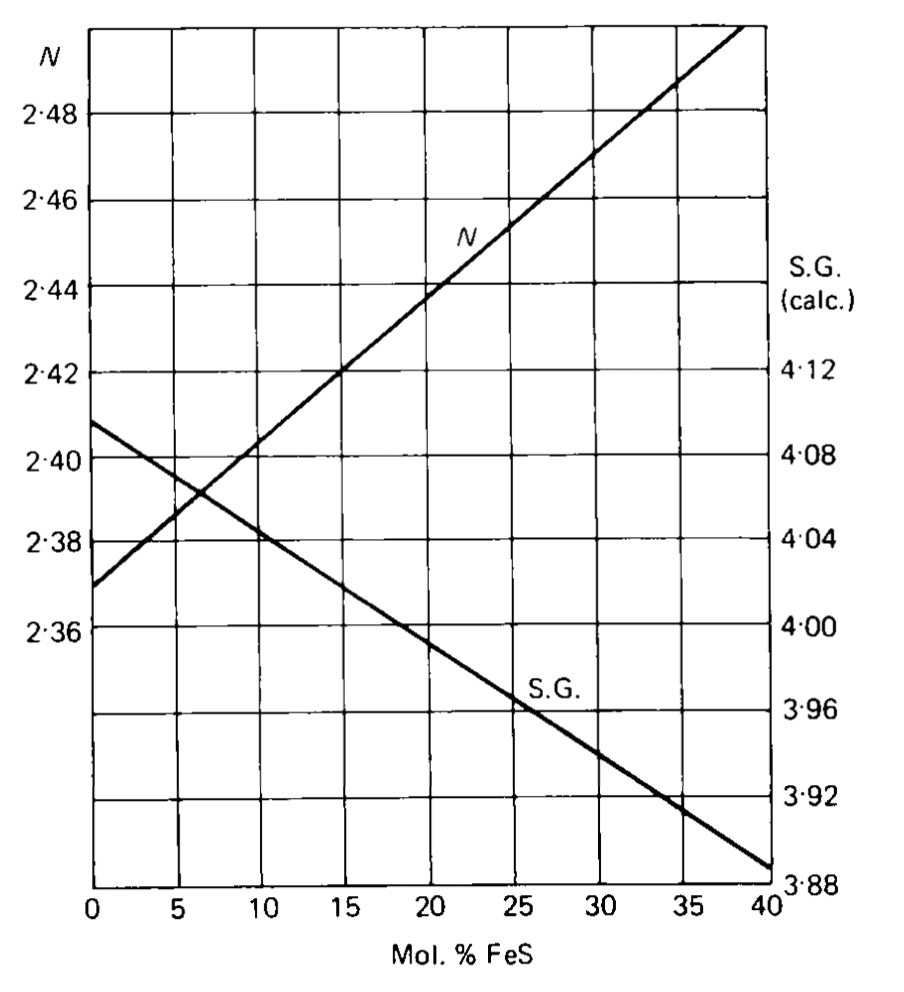
Refractive index and specific gravity in sphalerites may vary due to variable chemical composition.
Since sphalerite ranks as the primary ore for zinc mining, scientists have synthesized this mineral for industrial research. In addition, facetable synthetic sphalerites in all their colors have appeared in jewelry and gem collections.
There are no known gemstone treatments for sphalerites.
Sphalerites don't normally display cat's eye or star stone effects. However, in a 2009 study, researchers Hyršl and Steinbach found that polishing scratches on the bottoms of sphalerite cabochons may cause apparent chatoyancy and asterism. In this case, these scratches may have occurred accidentally. (pp. 303-304)
Significant gem-quality sources include the following:

From the large, reddish material from Spain, faceters could easily cut gems of hundreds of carats, as well as cabochons. Green cleiophane material from New Jersey has yielded faceted gems as large as 15 carats. Mexican material could yield faceted gems to 50 carats.

Clean sphalerites only with a soft brush, mild detergent, and warm water. See our gemstone jewelry cleaning guide for more recommendations.
Hoffmann, C. and U. Henn, reviewed by Mahinda Gunawardene. "Grüne Sphalerite aus Zaire (Green sphalerite from Zaire)." Gemological Abstracts. Gems & Gemology, Spring 1985. (Accessed 4/25/24)
Hyršl, Jaroslav and Martin P. Steinbach. "Pseudo-chatoyancy and pseudo-asterism in sphalerite from Spain." Gems & Gemology, Winter 2009. (Accessed 4/25/24)









When you join the IGS community, you get trusted diamond & gemstone information when you need it.

Radiant square-cut sphalerite, 198 cts, 30.1 x 30.3 x 21.1 mm, Las Manforas, Aliva, Picos de Europa, Spain.
Sphalerites have low hardness (3.5 to 4) and perfect cleavage, which make them less than ideal as jewelry stones. Nevertheless, sphalerites can show beautiful colors and may appeal to collectors of unusual gems. Use protective settings for these gems, especially if worn as ring stones.
For custom cutting, you can easily find facetable sphalerite rough in most colors. Faceting this material, however, poses a challenge. Consult our sphalerite buying guide for more information.

Sphalerites come in many colors, including brown, orange, yellow, green, orange-red, red, whitish gray, black, and even (very rarely) colorless. Sphalerites can also show color zoning.

According to a 1984 study by Hoffmann and Henn, a number of trace elements may contribute to color in sphalerites. These include germanium, calcium, copper, mercury, and cerium (yellow); tin, silver, and molybdenum (reddish); and cobalt and iron (green). (p. 49) Iron-rich sphalerites appear black.

Iron-rich, opaque, black stones are known as marmatites, named after the locality, Marmato, Italy.
In Europe, sphalerite is sometimes called blende, after the German word blenden, "to dazzle." Though they have no connection to actual ruby gems, stones with red and orange shades are sometimes called "ruby blende."

Low-iron, pale and colorless sphalerites, known as cleiophanes, are extremely rare.

Sphalerites have an exceptionally high dispersion of 0.156. Well-cut sphalerites can display a beautiful rainbow effect of multi-colored flashes of light. Only other rarely faceted gemstones have comparable dispersion values. Gemologists can measure a gem's dispersion using a refractometer or a spectrometer. However, keep in mind that cut quality, color, and specific gravity can affect how well a specimen displays dispersion.

Sphalerite and wurtzite are polymorphs. They share the same chemical formula (ZnS), but have different crystal habits. Sphalerite has an isometric crystal structure, while wurtzite has a hexagonal structure. These two minerals can coexist in alternating layers as schalenblende, a gem material used rarely for cabochons.

Sphalerites may have a streak color ranging from pale brown to yellow or white. Keep in mind that streak testing can destroy the test sample. Conduct this examination only as a last resort for gem identification on rough, never on a finished gem. This test will also release a sulfurous odor from the test sample.
Sphalerites are pyroelectric. When heated, these gems generate an electrical charge.
Sphalerites may show anomalous double refraction due to crystal strain.
Refractive index and specific gravity in sphalerites may vary due to variable chemical composition.

Since sphalerite ranks as the primary ore for zinc mining, scientists have synthesized this mineral for industrial research. In addition, facetable synthetic sphalerites in all their colors have appeared in jewelry and gem collections.
There are no known gemstone treatments for sphalerites.
Sphalerites don't normally display cat's eye or star stone effects. However, in a 2009 study, researchers Hyršl and Steinbach found that polishing scratches on the bottoms of sphalerite cabochons may cause apparent chatoyancy and asterism. In this case, these scratches may have occurred accidentally. (pp. 303-304)
Significant gem-quality sources include the following:

From the large, reddish material from Spain, faceters could easily cut gems of hundreds of carats, as well as cabochons. Green cleiophane material from New Jersey has yielded faceted gems as large as 15 carats. Mexican material could yield faceted gems to 50 carats.

Clean sphalerites only with a soft brush, mild detergent, and warm water. See our gemstone jewelry cleaning guide for more recommendations.
Hoffmann, C. and U. Henn, reviewed by Mahinda Gunawardene. "Grüne Sphalerite aus Zaire (Green sphalerite from Zaire)." Gemological Abstracts. Gems & Gemology, Spring 1985. (Accessed 4/25/24)
Hyršl, Jaroslav and Martin P. Steinbach. "Pseudo-chatoyancy and pseudo-asterism in sphalerite from Spain." Gems & Gemology, Winter 2009. (Accessed 4/25/24)
Sphalerite occurs in many colors, including green, yellow, orange, brown, and fiery red. With a dispersion over three times that of diamond and an adamantine luster, faceted specimens make beautiful additions to gem collections. However, they’re too soft for most jewelry uses.
